First blog post in
over five years! Do people even read blogs anymore? Other than when they maybe
pop up in a Google search? If not, welcome fellow googlers!
This
disease-related post is a little more personal than my other
illness/disease-related posts. In 2017, I had an incidental (happenstance)
finding on an MRI that I had done due to recurring migraines and some
peripheral neuropathy (numbness and tingling in the limbs). There were no signs
of any injury or damage that could explain the neuropathy, but they did find a
small acoustic neuroma, aka vestibular schwannoma, which is a
benign/non-cancerous tumor on the vestibular nerve. The name acoustic neuroma is
a misnomer because the tumors are most often found on the vestibular nerve,
which is the cranial nerve that helps control balance. This type of tumor forms
due to an overproduction of the cells that wrap around and insulate the nerve,
called Schwann cells.

The vestibular nerve and cochlear nerve (aka acoustic nerve, for hearing) are between the machinery of the ear and the brain. They come together between the brain and the ear and then split apart again after going through the skull. These nerves travel through the internal auditory canal, which is like a small hallway in the skull, with the facial nerve. Acoustic neuromas often develop on the section of the vestibular nerve in this canal.
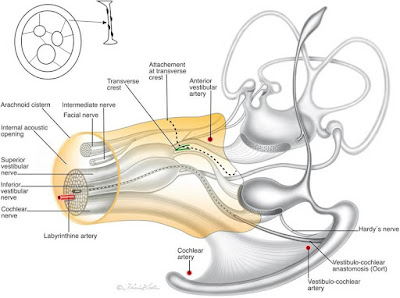 |
| The internal auditory canal, a hallway in the skull, and surrounding structures. Credit: Mei et al., 2019. |
Approximately 1-2 in 100,000 people have an acoustic neuroma and in 95% of cases they’re on one side only. In about 5% of cases, a tumor will develop on both sides. These are associated with a genetic disorder called neurofibromatosis type 2. Approximately 70% of diagnosed acoustic neuromas won't continue to grow and can be monitored via a watch-and-wait approach to confirm the lack of growth. This entails repeat MRIs at regular intervals, usually once a year. If there is no growth and there are no symptoms they can be left alone.
 |
| Credit: Slide-to-Doc.com |
If this type of tumor grows large enough, it can protrude out of the internal auditory canal toward the brain and, because there is only so much room in the skull, they press on other brain structures, like the cerebellum and brain stem, which can cause additional symptoms and eventually even be fatal though this is very rare.
For acoustic neuromas that are actively growing, there are two major courses of treatment: radiation or surgery. But there are different approaches both for radiation and surgery and the choice of which to pursue is dependent on many factors, such as the patient's age, the size and location of the tumor, and the extent of symptoms being experienced. In general, the the decision about which treatment option to pursue is very personal and I can only present to you what I learned in the course of my own personal experience.
My first thought upon being diagnosed was, "I'm glad it's not cancer." This was quickly followed by relief that it was found when it was still very small and when I was not yet experiencing symptoms. At that time, and as of last July, I still had perfect hearing in both ears and only an occasional weird feeling of fullness or pressure in my affected ear. Almost like how it feels before your ears pop on an airplane but just out of the blue and it doesn't go away with the usual tricks to pop your ears. Because of the size and lack of symptoms, we decided to watch-and-wait.
I had another MRI six months later and then another 12 months after that and another 12 months later. Due to the small size and variations in MRI images it was difficult for my otolaryngologist (ear, nose, and throat aka ENT) doctor to tell if it was growing or if it was just normal variation. He consulted with a group of experts and they concluded that it was indeed growing but recommended waiting for one more MRI before making any major decisions. That MRI would have been done in April 2020 but we all know what happened in early 2020. After my MRI in 2021, my doctor said it was good, in a way, that I skipped a year because it made the growth more obvious. He recommended radiation, so I made a consultation appointment for radiation.
The radiation used for acoustic neuromas, and I'm sure for brain tumors of any kind, is very precise to ensure that the tumor is irradiated but the surrounding brain tissue receives little to no radiation. GammaKnife radiation uses radioactive cobalt, is performed in one sitting, and usually utilizes a head frame affixed to the skull to keep the head stationary throughout the procedure. CyberKnife uses a stream of photos and the procedure is usually accomplished over several sessions and utilizes a hard, mesh face mask that is screwed into the table to keep the head stationary. Initially, I thought GammaKnife was my best option, however, the Acoustic Neuroma Association (ANA) recommends having consultations for both radiation and surgery so I made appointments with some surgeons.
The surgical options for acoustic neuromas come in three flavors: translabyrinthine, retrosigmoidal, and middle fossa. Each approach has a "best fit" for certain profiles. For people who have already lost all serviceable hearing in the affected ear, the translabyrinthine approach is a good option. This is because this approach goes at the tumor from an angle that provides an excellent view to dissect the tumor and avoid the facial nerve but does not preserve their hearing and, in fact, destroys any remaining hearing. The retrosigmoidal approach is "versatile" according to the scientific literature, meaning it can be a good approach for various tumor sizes and positions and it is possible to preserve hearing, but it does have a slightly higher risk of certain side effects than the middle fossa approach. The middle fossa approach provides the best possibility of preserving serviceable hearing and is a good option for people who still have great hearing. This is the treatment option recommended to me by every surgeon I spoke to, even those whose facilities do both radiation and surgery.
 |
| Credit: Slide-to-Doc.com |
Initially, I thought GammaKnife was the treatment for me. But after I spoke with some surgeons, including a facility that does both surgery and radiation, I decided a good ol' fashioned dive into the scientific literature was going to be my decision maker. Radiation was touted to me as the best option because it's non-invasive (I mean, relatively speaking) and has the best potential to preserve my hearing. But the literature showed that it's a little murkier than that. To understand, it's helpful to know how radiation works for acoustic neuromas.
Radiation is performed in either one sitting or several sessions, as mentioned previously, but there are no immediate effects. The tumor cells die over time and, months after radiation, hearing, balance, and, rarely, the facial nerve are affected. What I didn’t like about this option for myself was that you don’t know when the balance and hearing issues will start, how bad they’ll be or get, or how long they’ll last. In addition, the preservation of hearing is... not necessarily true. For many patients, the hearing loss continues even after all the tumor cells are dead. Within 10 years of radiation for an acoustic neuroma, most people have lost serviceable hearing. There are some interesting hypotheses about this regarding the inflammation caused by the death of the tumor cells but I’m not going to go deep into that. Suffice it to say, radiation is a great option for people who have already lost hearing, are older and losing hearing due to aging anyway, or people who just don't want surgery for whatever reason. Again, treatment decisions are highly personal! I didn't personally feel radiation was the best choice for me, which is why I ended up choosing the middle fossa surgical approach.
With surgery, the hearing you have after recovery from surgery is the hearing you're going to have. My surgeon estimates the likelihood of preserving serviceable hearing for me at slightly greater than 60%. While, as an audiophile, those odds kind of suck, I will take the chance to keep my hearing over having definitely worsening hearing over the next decade and being almost guaranteed single-sided deafness (SSD) at the end of it. Maybe I will lose all my hearing in that ear during surgery. That would suck, but at least I'll know and adapt immediately. Similarly, I will need to immediately adapt to deficits in my sense of balance.
Immediately after surgery, most people who haven't had many balance issues yet will have awful balance and walk around looking like their drunk at first. The vestibular nerve is cut during surgery to remove the tumor so the nerve on the other side of the head has to learn to compensate for the loss. Luckily, it does so fairly quickly and other cues, such as vision, can help compensate as well. Most people are back to work after 4-6 weeks and back to running after 4-12 weeks (based on anecdotal data from the ANA forums). No one I could find that had an acoustic neuroma is also an aerialist so I will have to find out how quickly I can get back to my silks classes myself. Everyone is different anyway. I do know that I, again, prefer the idea of getting it over with all at once to dealing with the worsening symptoms I would have after radiation. Aerial silks are my favorite hobby of all time and it would be very demoralizing to have worsening balance issues without knowing when they would end.
The last thing I preferred the surgical option for was the recovery time. I’ll be able to take time off from work to rest, recover, and work on my balance instead of working through worsening hearing and balance and the mental anguish I know that would cause me if I chose radiation.
This was a bit of a ramble but if you're still reading: if you only had about ten days until you could possibly only hear out of one ear, what is the one song you would absolutely have to listen to one more time?




